Haemoglobin is the molecule responsible for transporting oxygen around the body. It is found in red blood cells. It picks up oxygen in the lungs, transports it through the blood and releases it in the tissues. In a fetus, haemoglobin picks up oxygen in the placenta, where it diffuses across the placenta from the mother’s haemoglobin.
Fetal Versus Adult Haemoglobin
Haemoglobin is formed of two pairs of protein subunits:
- Fetal haemoglobin (HbF) has two alpha and two gamma subunits (α₂γ₂)
- Adult haemoglobin (HbA) has two alpha and two beta subunits (α₂β₂)
Fetal haemoglobin has a greater affinity for oxygen than adult haemoglobin. Oxygen binds to fetal haemoglobin more easily and dissociates (separates) less easily. This allows fetal haemoglobin to “steal” oxygen from the mother’s haemoglobin in the placenta. If fetal and maternal haemoglobin had the same oxygen affinity, oxygen transfer to the fetus would be less efficient.
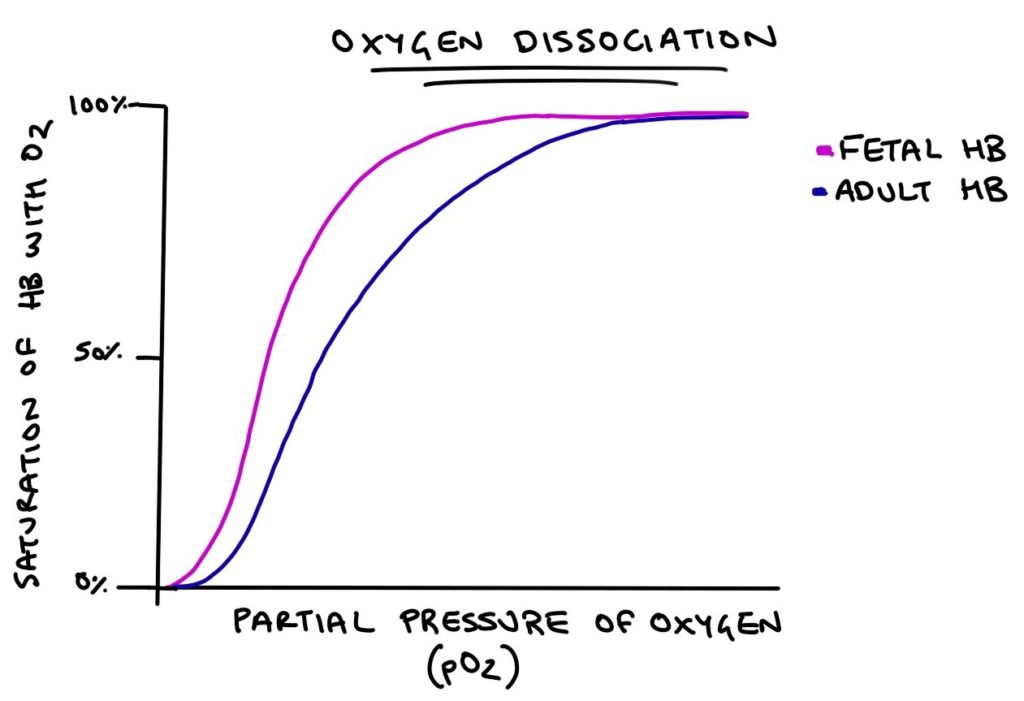
Oxygen Dissociation Curve
The oxygen dissociation curve shows the affinity of fetal and adult haemoglobin for oxygen. Along the x-axis is the partial pressure of oxygen (pO₂), which reflects the oxygen concentration in the blood. On the y-axis is the percentage saturation of haemoglobin with oxygen (how “full” the haemoglobin molecule is with oxygen).
As the partial pressure of oxygen increases, more oxygen will be bound to haemoglobin. Adult haemoglobin requires a higher partial pressure of oxygen to achieve the same level of saturation as fetal haemoglobin.
At Birth
From 32 to 36 weeks of gestation, production of HbF decreases. At the same time, HbA is produced in greater quantities. Over time, there is a gradual transition from HbF to HbA.
At birth, around 70–80% is HbF and 20–30% is HbA. By 6 months, very little HbF is produced, and red blood cells contain almost entirely HbA.
Last updated March 2026
Now, head over to members.zerotofinals.com and test your knowledge of this content. Testing yourself helps identify what you missed and strengthens your understanding and retention.

