Myasthenia gravis is an autoimmune condition affecting the neuromuscular junction. It causes muscle weakness that progressively worsens with activity and improves with rest.
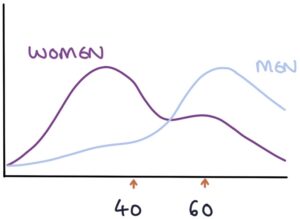
Myasthenia gravis affects men and women at different ages, typically affecting women under 40 and men over 60.
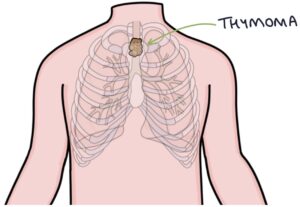
There is a strong link with thymomas (thymus gland tumours). 10-20% of patients with myasthenia gravis have a thymoma. 30% of patients with a thymoma develop myasthenia gravis.
Pathophysiology
Motor neurones communicate with muscles via the neuromuscular junction. On one side of the synapse is the presynaptic membrane of the axon terminal of the motor neurone. On the other side is the postsynaptic membrane of the motor end plate of the muscle cell.
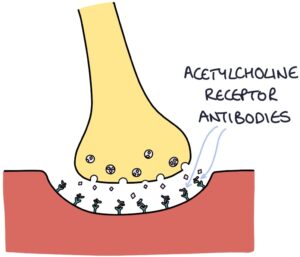
The axons release a neurotransmitter called acetylcholine from the presynaptic membrane. Acetylcholine travels across the synapse and attaches to receptors on the postsynaptic membrane, simulating muscle contraction.

Acetylcholine receptor (AChR) antibodies are found in most patients with myasthenia gravis. These antibodies bind to the postsynaptic acetylcholine receptors, blocking them and preventing stimulation by acetylcholine. The more the receptors are used during muscle activity, the more they become blocked. There is less effective stimulation of the muscle with increased activity. With rest, the receptors are cleared, and the symptoms improve.
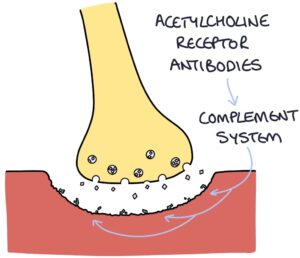
These antibodies also activate the complement system within the neuromuscular junction, leading to cell damage at the postsynaptic membrane, further worsening symptoms.
Two other antibodies can cause myasthenia gravis:
- Muscle-specific kinase (MuSK) antibodies
- Low-density lipoprotein receptor-related protein 4 (LRP4) antibodies
MuSK and LRP4 are important proteins for the creation and organisation of the acetylcholine receptor. Destruction of these proteins leads to inadequate acetylcholine receptors.
Presentation
Symptoms vary dramatically between patients, ranging from mild to life-threateningly severe. The critical feature is weakness that worsens with muscle use and improves with rest. Symptoms are typically best in the morning and worst at the end of the day.
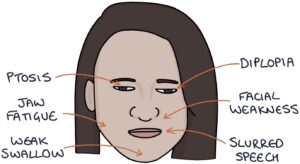
The symptoms most affect the proximal muscles of the limbs and small muscles of the head and neck, with:
- Difficulty climbing stairs, standing from a seat or raising their hands above their head
- Extraocular muscle weakness, causing double vision (diplopia)
- Eyelid weakness, causing drooping of the eyelids (ptosis)
- Weakness in facial movements
- Difficulty with swallowing
- Fatigue in the jaw when chewing
- Slurred speech
There are a few ways to elicit fatiguability in the muscles:
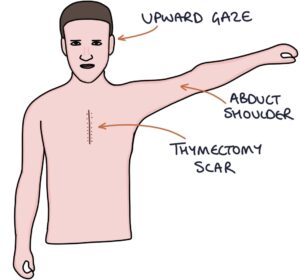
- Repeated blinking will exacerbate ptosis
- Prolonged upward gazing will exacerbate diplopia on further testing
- Repeated abduction of one arm 20 times will result in unilateral weakness when comparing both sides
Further examination steps include:
- Checking for a thymectomy scar
- Testing the forced vital capacity (FVC)
Investigations
Antibody tests look for:
- AChR antibodies (around 85%)
- MuSK antibodies (less than 10%)
- LRP4 antibodies (less than 5%)
A CT or MRI of the thymus gland is used to look for a thymoma.
The edrophonium test can be helpful where there is doubt about the diagnosis.
Edrophonium Test
Patients are given intravenous edrophonium chloride (or neostigmine). Normally, cholinesterase enzymes in the neuromuscular junction break down acetylcholine. Edrophonium blocks these enzymes, reducing the breakdown of acetylcholine. As a result, the level of acetylcholine at the neuromuscular junction rises, temporarily relieving the weakness. A positive result suggests a diagnosis of myasthenia gravis.
Treatment options
Treatment options include:
- Pyridostigmine is a cholinesterase inhibitor that prolongs the action of acetylcholine and improves symptoms
- Immunosuppression (e.g., prednisolone or azathioprine) suppresses the production of antibodies
- Thymectomy can improve symptoms, even in patients without a thymoma
- Rituximab (a monoclonal antibody against B cells) is considered where other treatments fail
Myasthenic Crisis
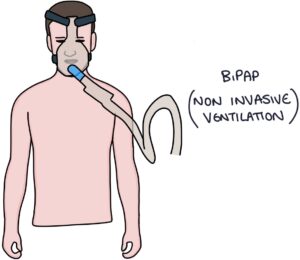
Myasthenic crisis is a potentially life-threatening complication of myasthenia gravis. It causes an acute worsening of symptoms, often triggered by another illness, such as a respiratory tract infection. Respiratory muscle weakness can lead to respiratory failure. Patients may require non-invasive ventilation or mechanical ventilation.
Treatment is with IV immunoglobulins and plasmapheresis.
Last updated October 2023
Now, head over to members.zerotofinals.com and test your knowledge of this content. Testing yourself helps identify what you missed and strengthens your understanding and retention.

