Syndrome of inappropriate antidiuretic hormone (SIADH) refers to the increased release of antidiuretic hormone (ADH) from the posterior pituitary. This increases water reabsorption from the urine, diluting the blood and leading to hyponatraemia (low sodium).
Pathophysiology
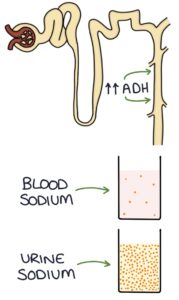
Antidiuretic hormone (ADH) is produced in the hypothalamus and secreted by the posterior pituitary gland. It is also known as vasopressin. ADH stimulates water reabsorption from the collecting ducts in the kidneys.
There are two potential sources of too much ADH:
- Increased secretion by the posterior pituitary
- Ectopic ADH, most commonly by small cell lung cancer
Excessive ADH results in increased water reabsorption in the collecting ducts, diluting the blood. This excess water reduces the sodium concentration (hyponatraemia). The extra water is not usually significant enough to cause fluid overload. SIADH results in euvolaemic hyponatraemia. Euvolaemic means normal (eu-) volume (-vol-) of blood (-aemic).
The urine becomes more concentrated as the kidneys excrete less water. Therefore patients with SIADH have high urine osmolality and high urine sodium.
Presentation
The symptoms of SIADH relate to low sodium (hyponatraemia). Depending on the sodium level and how rapidly it occurs, they may be asymptomatic or present with non-specific symptoms:
- Headache
- Fatigue
- Muscle aches and cramps
- Confusion
Severe hyponatraemia can cause seizures and reduced consciousness.
Causes
There is a long list of causes of SIADH:
- Post-operative after major surgery
- Lung infection, particularly atypical pneumonia and lung abscesses
- Brain pathologies, such as a head injury, stroke, intracranial haemorrhage or meningitis
- Medications (e.g., SSRIs and carbamazepine)
- Malignancy, particularly small cell lung cancer
- Human immunodeficiency virus (HIV)
TOM TIP: The top three causes of SIADH to remember are post-operative, SSRIs and small cell lung cancer. I have seen several cases of SIADH relating to major surgery and the use of SSRIs. SIADH relating to small cell lung cancer is an exam favourite.
Diagnosis
There is no single test to diagnose SIADH. The diagnosis is based on the clinical features:
- Euvolaemia
- Hyponatraemia
- Low serum osmolality
- High urine sodium
- High urine osmolality
Other causes of hyponatraemia need to be excluded:
- Short synacthen test to exclude adrenal insufficiency
- No history of diuretic use
- No diarrhoea, vomiting, burns, fistula or excessive sweating
- No excessive water intake
- No chronic kidney disease or acute kidney injury
- No heart failure or liver disease
Primary polydipsia involves excessive water consumption with no underlying cause, diluting the blood and urine. This also causes euvolaemic hyponatraemia. However, there is a low urine sodium and urine osmolality.
Establish the Cause
Sometimes the cause will be obvious, for example, in someone that recently started an SSRI, has a chest infection or recent major surgery. The cause is confirmed if the hyponatraemia resolves when the cause is removed.
A chest x-ray can be used to assess for pneumonia, lung abscess and lung cancer.
When no clear cause can be found, malignancy should be excluded. The NICE clinical knowledge summaries (updated 2020) suggest considering a CT thorax, abdomen and pelvis and MRI head to find the malignancy.
Management
Management involves:
- Admission if symptomatic or severe (e.g., sodium under 125 mmol/L)
- Treating the underlying cause (e.g., stopping causative medications or treating the infection)
- Fluid restriction
- Vasopressin receptor antagonists (e.g., tolvaptan)
It is essential to correct the sodium slowly to prevent osmotic demyelination. The sodium concentration should not change more than 10 mmol/L in 24 hours.
Fluid restriction involves limiting the patient’s fluid intake to 750-1000 ml per day. This may be enough to correct hyponatraemia without the need for medications.
Vasopressin receptor antagonists, such as tolvaptan, work by blocking ADH receptors. They can cause a rapid rise in sodium. They are initiated by an endocrinologist and require close monitoring (e.g., 6 hourly sodium levels).
Osmotic Demyelination Syndrome
Osmotic demyelination syndrome is also known as central pontine myelinolysis (CPM). It is usually a complication of long-term severe hyponatraemia (e.g., under 120 mmol/L) being treated too quickly (e.g., more than a 10 mmol/L increase per 24 hours).
As the blood sodium concentration drops, water will move by osmosis across the blood-brain barrier into the cells of the brain. Water moves from the area of low concentration of solutes (the blood) to the area of high concentration of solutes (the brain). This causes the brain to swell with fluid. The brain adapts to this by reducing the solutes in the brain cells so that water is balanced across the blood-brain barrier and the brain does not become oedematous. This adaptation takes a few days. Therefore, if hyponatraemia has been present and severe for a long time, the brain cells will also have a low osmolality. This is only a problem once the blood sodium levels rapidly rise, causing water to rapidly shift out of the brain cells and into the blood. This causes two phases of symptoms.
The first phase is due to the electrolyte imbalance. The patient presents as encephalopathic and confused. They may have a headache, vomiting and seizures. These symptoms often resolve before the onset of the second phase.
The second phase is due to the demyelination of the neurones, particularly in the pons. This occurs a few days after the rapid correction of sodium. This may present with spastic quadriparesis, pseudobulbar palsy and cognitive and behavioural changes. There is a significant risk of death.
Prevention is essential as treatment is only supportive once osmotic demyelination occurs. A proportion of patients make a clinical improvement, but most are left with some neurological deficit.
Last updated March 2023
Now, head over to members.zerotofinals.com and test your knowledge of this content. Testing yourself helps identify what you missed and strengthens your understanding and retention.

